PFACs are valuable partners with researchers, however a PFAC’s role in research projects has to be defined by its preference for the level of engagement and is typically limited by the PFAC’s other priorities. This doesn’t mean that activities PFACs can participate in won’t yield meaningful results for research efforts. It just demands that researchers are thoughtful in planning how they want to engage PFACs.
Examples of Research Activities for PFACs
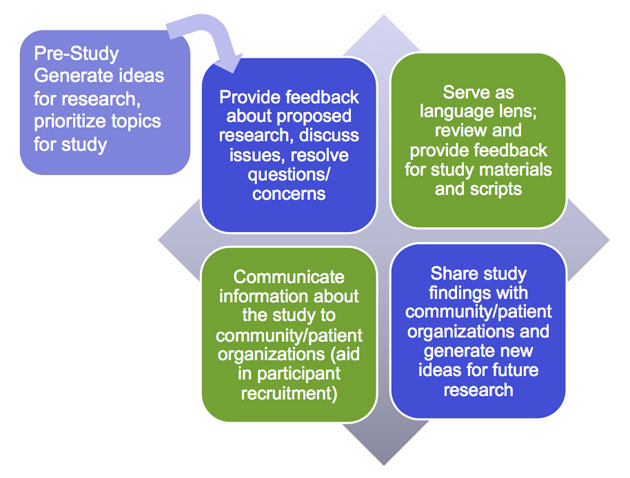
Often, PFACs are asked to participate in a perfunctory, less meaningful way than they are capable of or desire. For example, some report that researchers present project findings and dissemination plans to them without any prior PFAC involvement or the PFAC is asked to provide feedback on materials that are at a final stage. Because PFACs are experienced in partnering with health care professionals, if given the opportunity they can help shape a research project in all stages from pre-planning to dissemination.
The following chart details potential activities that PFACs can participate in within the different stages. In the right hand column are questions that PFACs and researchers can use to spark rich and purposeful discussions.
Activities and Questions for PFACs in Stages of Research
| Stages | PFAC Potential Activities | Questions for Advisors |
|---|---|---|
| Pre-Planning |
|
|
| Planning |
|
|
| Pilot |
|
Note: Questions listed above in the Planning Stage are appropriate to use in this stage. |
| Data Collection and Enrollment |
|
|
| Analysis |
|
|
| Dissemination |
|
|

